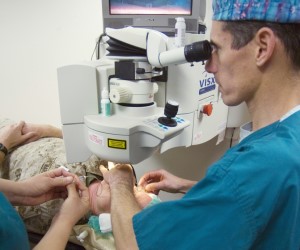
Industry groups challenge FDA’s LASIK guidance, call for withdrawal
 Advertising, Promotion and LabelingComplianceHealth Authority meeting and communication strategyMedical DevicesNorth AmericaProduct LifecycleRegulatory Intelligence/Policy
Advertising, Promotion and LabelingComplianceHealth Authority meeting and communication strategyMedical DevicesNorth AmericaProduct LifecycleRegulatory Intelligence/Policy Advertising, Promotion and LabelingComplianceHealth Authority meeting and communication strategyMedical DevicesNorth AmericaProduct LifecycleRegulatory Intelligence/Policy
Advertising, Promotion and LabelingComplianceHealth Authority meeting and communication strategyMedical DevicesNorth AmericaProduct LifecycleRegulatory Intelligence/Policy