Advance your career with industry-recognized credentials
Demonstrate expertise. Differentiate yourself. Drive impact.
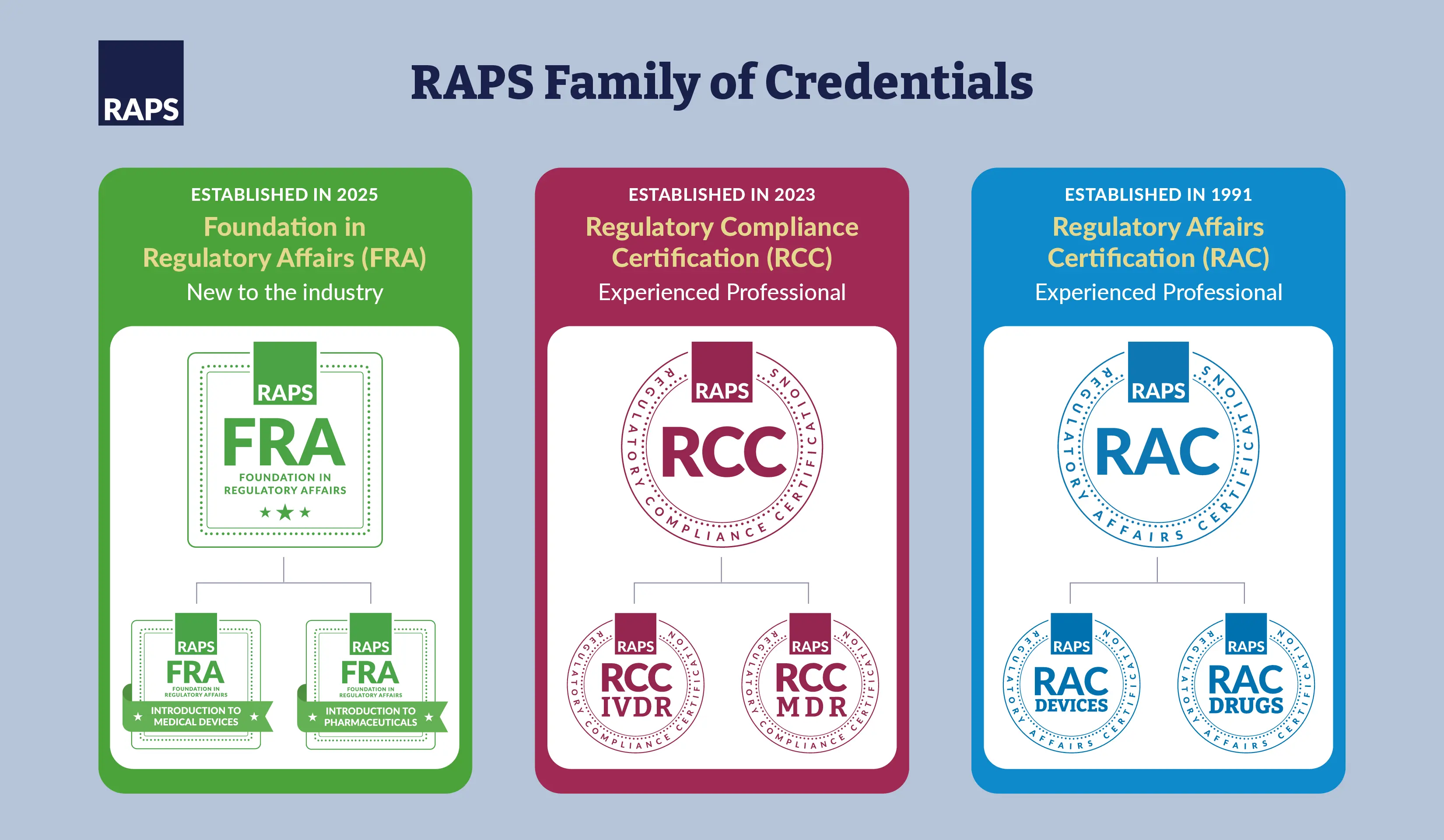
Whether you are new to regulatory affairs or a seasoned professional looking to stand out, RAPS offers three globally recognized credentials designed to validate your knowledge and advance your career.

The Regulatory Affairs Certification (RAC) is the leading credential for regulatory professionals in the healthcare sector. The two RAC designations are Pharmaceuticals (RAC-Drugs) and Medical Devices (RAC-Devices). The RAC is intended for individuals employed in regulatory agencies, industry, consultancies and other settings involved with the regulation of healthcare products.
APPLY FOR AN EXAM

The Regulatory Compliance Certifications are credentials designed by regulatory professionals for regulatory professionals. There are two RCC exams: The RCC-IVDR and the RCC-MDR. This designation provides third-party validation, indicating that a certification holder understands European regulations for in vitro diagnostics (IVDs) or medical devices.
APPLY FOR AN EXAM

Show the world that you have the foundational knowledge regulatory affairs professionals need. The new Foundation in Regulatory Affairs (FRA) assessment-based certificate is designed to help you build regulatory affairs knowledge and then test that knowledge through an exam.
APPLY FOR AN EXAM
Why Earn a RAPS Credential
RAPS credentials are recognized globally across pharmaceuticals, biologics, medical devices, and related life sciences sectors. They help you:
- Demonstrate verified regulatory or compliance competence
- Strengthen your credibility with employers and peers
- Position yourself for new roles and promotions
- Build confidence navigating complex regulatory frameworks
- Join a global community of recognized regulatory professionals
RAC vs. RCC. vs. FRA: What’s the Difference?
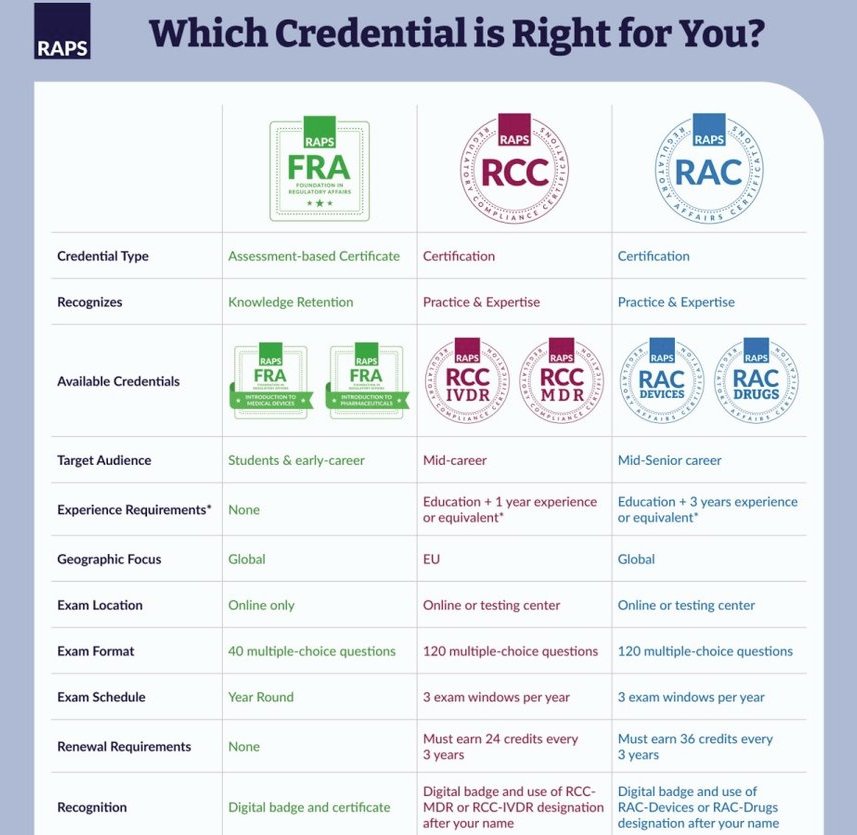
*The eligibility requirements for the RCC and RAC are more detailed than what this chart shows. Please see our website for full eligibility details
Find your Fit
Foundational Knowledge: Foundation in Regulatory Affairs (FRA)
The FRA is an assessment-based certificate that helps you prove you have the knowledge to start and succeed in regulatory affairs.
Ideal for:
- Students
- Entry-level professionals (0-2 years of experience)
- Individuals transitioning into regulatory affairs from other fields (e.g., quality, clinical, pharmacy, R&D)
Why earn the FRA
- Demonstrate a solid understanding of regulatory basics to employers
- Provide a structured path for learning essential concepts
- Boost confidence and credibility in an entry-level role
Build Your Foundation
Earning an FRA credential demonstrates fundamental proficiency that could be helpful in securing a position in the regulatory field.
Michelle Lott, RAC
Principal and Founder, leanRAQA
Specialized Expertise: The Regulatory Compliance Certification (RCC)
The RCC validates your EU MDR and EU IVDR knowledge.
Ideal for:
- Mid-career professionals who are PRRCs or specialize in EU compliance
- Professionals in quality assurance or other roles that require deep knowledge of EU regulations
Why earn the RCC
- Demonstrate to evaluators that you meet the MDR and IVDR PRRC requirements.
- Validate current, specialized knowledge of a critical and complex regulatory area.
- Help companies ensure compliance and mitigate risk in the EU market
- Add a highly marketable and specific certification to your resume to increase your employment options and compensation negotiating power
Having a candidate delve deep into a regulation and testing that knowledge would give any employer confidence that the candidate knows what they are talking about.
Mastery: The Regulatory Affairs Certification (RAC)
The RAC is the premier, globally recognized certification for regulatory professionals in the healthcare products sector.
Ideal for:
- Mid to senior level professionals, consultants, and directors
- Those who manage products globally or have a broad scope of regulatory responsibilities
Why earn the RAC:
- Elevate your professional credibility on a global scale
- Validate your ability to apply regulatory knowledge across the entire product lifecycle, validate breadth and strategical application
- Increased earning potential (12% higher for RAC holders according to 2024 RAPS scope of practice survey)
It really demonstrates a level of commitment to oneself and one's career, and as a hiring manager, it's definitely something I look for and something I'd encourage.
Paul Swift
Carl Zeiss Meditec
RAPS Certificates and Certifications: How to Pick the Right One
Learn the key difference between a certificate and a certification and which option is right for you now and in the future.








