About RAPS
Mission
Develop and sustain a highly competent global regulatory workforce that drives good regulatory practice and policy to advance public health.

The Regulatory Affairs Professionals Society (RAPS) is the largest global organization of professionals involved with regulatory and quality for healthcare products, including medical devices, pharmaceuticals and biologics, diagnostics, and digital health. Founded in 1976 as a neutral, nonprofit organization, RAPS supports and elevates the regulatory profession with education and training, professional standards, publications, research, networking, career development, and other valuable resources. RAPS Institute is an affiliated organization that is home to the Regulatory Affairs Certifications (RAC) and the Regulatory Compliance Certifications (RCC), the only post-academic professional designations to recognize regulatory excellence in the healthcare products sector. The society is headquartered in suburban Washington, D.C., with chapters and affiliates worldwide. www.raps.org.
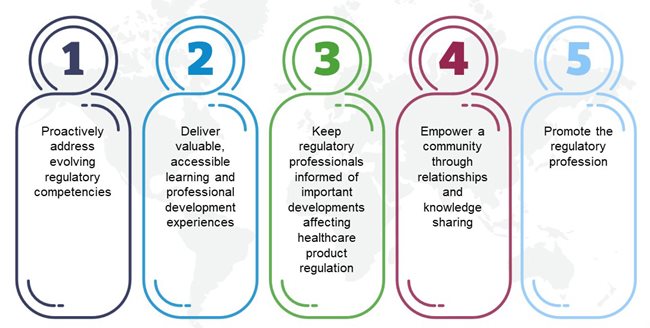
Strategic Priorities
RAPS by the numbers





