CDER lays out 2021 guidance agenda
The US Food and Drug Administration’s (FDA) Center for Drug Evaluation and Research (CDER) released a list of the new and revised draft guidances it plans to release in 2021.The agenda, while oftentimes aspirational, provides insights as to what the agency’s drug center’s new guidance priorities are for the coming year. However, the list does not include draft or revised guidances the agency plans to finalize in 2021. (RELATED: 2020 CDER draft and revised guidance: What’s coming, Regulatory Focus 30 January 2020).
Many of the planned guidances on the list were included in previous years’ agendas, such as the proposed clinical pharmacology draft guidance “Assessing the Effects of Food on Drugs in INDs or NDAs – General Considerations”, and updated guidance on “Pharmacogenomic Data Submission” and “Pharmacokinetics in Patients with Impaired Renal Function – Study Design, Data Analysis and Impact on Dosing and Labeling”.
In total, there are 105 guidances included on the agenda, 42 of which are new for 2021. The new guidances not carried over from 2020 are presented in the table at the bottom of the article. This year there are four new categories of draft guidances that appear on the agenda that were not included in 2020: animal rule, biosimilars, compounding and pharmacology/toxicology.
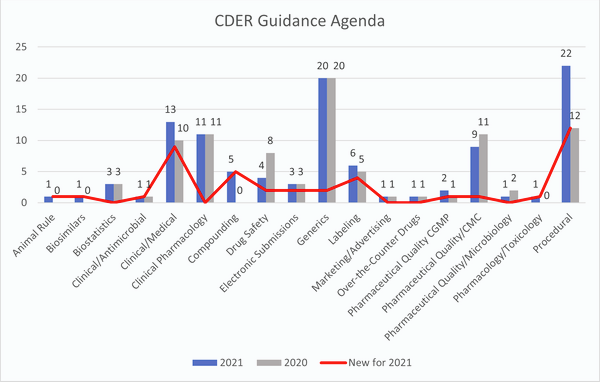
Some of the notable new guidances include a guidance on meeting the standard for substantial evidence based on a single adequate and well-controlled clinical trial plus confirmatory evidence and a guidance aimed at class-specific recommendations for biosimilars and interchangeable products. Several of the guidances on the agenda are aimed at providing recommendations for individualized antisense oligonucleotides (ASO), including guidance on clinical recommendations to support investigational new drug application (IND) submissions, nonclinical testing, and on chemistry, manufacturing and controls (CMC) considerations for the so-called “n of 1” therapies. (RELATED: ‘N of 1' therapies addressed in draft FDA guidance, Regulatory Focus 5 January 2021).
| Planned new and revised CDER draft guidance for 2021 | |
| Category | Guidance |
| Animal Rule | Development of Drugs for Acute Radiation Syndrome: Questions and Answers |
| Biosimilars | Product Class-Specific Recommendations for Developing Biosimilar and Interchangeable Biological Products |
| Clinical/Antimicrobial | Rabies: Developing Monoclonal Antibody Cocktails for the Passive Immunization Component of Post-Exposure Prophylaxis |
| Clinical/Medical | Assessment of Adhesion for Topical and Transdermal Systems Submitted in New Drug Applications |
| Bowel Cleansing for Colonoscopy: Developing Drugs for Treatment – Efficacy and Safety Considerations for Developing New Products |
|
| Celiac Disease: Developing Drugs for Adjunctive Treatment to a Gluten Free Diet | |
| Chemotherapy-Induced Nausea and Vomiting: Developing Drugs for Prevention | |
| Clinical Recommendations to Support IND Submissions for Individualized Antisense Oligonucleotide Drug Products for Severely Debilitating or Life-Threatening Diseases |
|
| Crohn’s Disease: Developing Drugs for Treatment | |
| Inborn Errors of Metabolism That Use Dietary Management: Considerations for Optimizing and Standardizing Diet in Clinical Trials for Drug Product Development | |
| Meeting the Substantial Evidence Standard Based on One Adequate and Well-Controlled Clinical Investigation and Confirmatory Evidence | |
| Ulcerative Colitis: Developing Drugs for Treatment | |
| Compounding | Compounded Drug Products That Are Essentially Copies of a Commercially Available Drug Product Under Section 503A of the Federal Food, Drug, and Cosmetic Act; Revised Draft |
| Compounded Drug Products That Are Essentially Copies of Approved Drug Products Under Section 503B of the Federal Food, Drug, and Cosmetic Act; Revised Draft | |
| Hospital and Health System Compounding Under the Federal Food, Drug, and Cosmetic Act; Revised Draft | |
| Prohibition on Wholesaling Under Section 503B of the Federal Food, Drug, and Cosmetic Act | |
| Safety Considerations for Container Labels and Carton Labeling Design to Minimize Medication Errors – Guidance for Outsourcing Facilities Under Section 503B of the FD&C Act | |
| Drug Safety | Postmarketing Studies and Clinical Trials: Determining Good Cause for Noncompliance with Section 505(o)(3)(E)(ii) of the Federal Food, Drug, and Cosmetic Act |
| Sponsor Responsibilities – Safety Reporting Requirements and Safety Assessment for IND and Bioavailability/Bioequivalence Studies | |
| Electronic Submission | Electronic Submission of Expedited Safety Reports from IND-Exempt BA/BE Studies |
| NDC Assignment of Human Drugs including Biological Product | |
| Generics | Abbreviated New Drug Application Submissions – Cover Letters |
| Waivers for pH Adjusters in Drug Products Intended for Parenteral, Otic, and Ophthalmic Use | |
| Labeling | Immunogenicity Information in Human Prescription Therapeutic Protein and Select Drug Product Labeling — Content and Format |
| Labeling for Biosimilar Products (Revision 1) | |
| Regulatory Considerations and Drug Labeling Recommendations for Prescription Drug Use-Related Software for Combination Products | |
| Statement of Identity and Strength — Content and Format of Labeling for Human Nonprescription Drug Products | |
| Pharmaceutical Quality CGMP | Non-Penicillin Beta-Lactam Drugs: A CGMP Framework for Preventing Cross-Contamination |
| Pharmaceutical Quality/CMC | Benefit-Risk Considerations for Product Quality Assessments |
| Pharmacology/Toxicology | Nonclinical Testing of Individualized Antisense Oligonucleotide Drug Products for Severely Debilitating or Life-Threatening Diseases |
| Procedural | Considerations for Rescinding Breakthrough Therapy Designation |
| Enhanced Drug Distribution Security at the Package Level Under the Drug Supply Chain Security Act | |
| Exclusivity for First Interchangeable Biological Product | |
| Expanded Access to Investigational Drugs for Treatment Use – Questions and Answers | |
| Fixed Dose Combinations and Single-Entity Versions of Previously Approved Antiretrovirals for the Treatment of Human Immunodeficiency Virus-1 Under President’s Emergency Plan for AIDS Relief (PEPFAR) | |
| Real-World Data: Assessing Electronic Health Records and Medical Claims Data to Support Regulatory Decision-Making for Drug and Biological Products | |
| Regulatory Considerations for the Use of Real-World Data and Real-World Evidence to Support Regulatory Decision-Making for Drugs and Biological Products | |
| Reporting Amount of Distributed Listed Drugs and Biological Products Under Section 510(j)(3) of the FD&C Act | |
| Responding to CGMP Observations on Form FDA 483 | |
| Use of Electronic Records and Electronic Signatures in Clinical Investigations Under 21 CFR Part 11 – Questions and Answers | |
| Using Registries as a Real-World Data Source for FDA Submissions | |
| Wholesale Distributor Verification Requirements for Saleable Returned Drug Product and Dispenser Verification Requirements When Investigating a Suspect or Illegitimate Product – Compliance Policy | |
FDA

