Eton’s Alkindi ads draw third OPDP untitled letter of 2021
The US Food and Drug Administration’s (FDA) Office of Prescription Drug Promotion (OPDP) last week sent an untitled letter to Eton Pharmaceuticals over sponsored links for the company’s pediatric adrenocortical insufficiency treatment Alkindi Sprinkle (hydrocortisone oral granules).OPDP said the sponsored links “are false or misleading in that they present information about the benefits of Alkindi Sprinkle, but fail to include any risk information about the drug,” thus rendering the product misbranded under the Federal Food, Drug, and Cosmetic Act.
In each of 10 example sponsored links for Alkindi Sprinkle released alongside the untitled letter, Eton promoted the drug for its intended use and highlighting its benefits, without mentioning its contraindications or other warnings and precautions related to the drug.
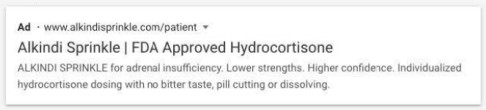
Other violative sponsored links included language such as “No cutting, no splitting, just sprinkles for neonates and children <17 years of age,” and “Alkindi Sprinkle is the accurate, hassle-free hydrocortisone granules for kids,” but without any corresponding risk information to satisfy fair balance requirements.
“These violations are especially concerning from a public health perspective because the promotional communications create a misleading impression about the safety of Alkindi Sprinkle, a drug that is used in a vulnerable pediatric patient population, and may cause serious adverse reactions such as adrenal crisis, infections, and growth retardation, among others,” OPDP wrote. Alkindi Sprinkle is contraindicated in patients who are hypersensitive to hydrocortisone and its label contains other warnings and precautions for decreased bone mineral density, psychiatric adverse reactions, and Cushing’s syndrome.
The untitled letter is the third sent by OPDP this year, after it sent letters to Amgen over a banner ad for its biological product Neulasta (pegfilgrastim) and Biohaven Pharmaceuticals for its migraine drug Nurtec (rimegepant). The office has also sent two warning letters in 2021, to AcelRX for its promotion of Dsuvia (sufentanil sublingual tablet) and CooperSurgical over a promotional video for its Paragard intrauterine device.
In response to the untitled letter, FDA has asked Eton for a reply within 15 business days to provide a plan for discontinuing any violative communications.
Untitled Letter, Promotional Materials




