MDSAP Sees Site Participation Increase
Major changes are afoot at the US Food and Drug Administration’s (FDA) Center for Devices and Radiological Health (CDRH) and a number of voluntary pilot programs for industry will be key in determining the direction they are heading, officials said at the 2018 FDA/Xavier MedCon conference on Thursday.
The Medical Device Single Audit Program (MDSAP) is among the voluntary programs that will help shape regulatory harmonization. Interest in the program, a pillar of the International Medical Device Regulators Forum (IMDRF), could benefit from more harmonization and has continued to gain momentum.
As illustrated in the graphic below, the auditing program has recently seen major spikes in the total number of participating manufacturer sites, totaling 1,749 in Q1 2018—up from just 788 in 2017.
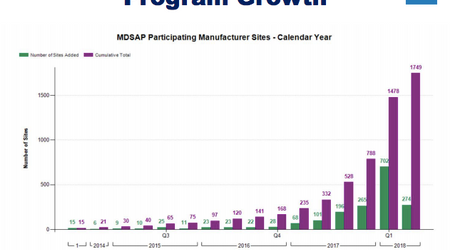
The growth in participation could be explained by Health Canada’s requirement that companies must comply with the program’s requirements by 1 January 2019, including the need for MDSAP audit reports to legally sell their devices in the Canadian market.
The set deadline has led to some scrambling among device makers to schedule audits in time, and despite the many tweaks implemented over the past year to facilitate the transition, many issues facing small- to medium-sized firms remain.
In contrast, device makers in the US have the option of using MDSAP audit reports as a substitute for routine surveillance inspections. Capt. Sean Boyd, deputy director for regulatory affairs at the center’s Office of Compliance, told Focus CDRH intends for MDSAP participation to remain optional in the US.
CDRH said its mission is to have 50% of firms of novel technologies come to the US in parallel with other major markets, if not first, by 2020, as described in a 3-year roadmap from January.
The relationship between these global efforts and extending the least burdensome principles of the FD&C Act relates to providing opportunities for device companies to satisfy regulatory requirements in multiple jurisdictions using mechanisms such as MDSAP, Boyd noted.
"Harmonization is an important aspect of least burdensome,” Boyd said. “MDSAP is a great example of a program where we're achieving that goal and we expect that to continue over the coming years."
Boyd pointed to other opportunities for expanding the single audit program, including the deployment of an information management system to facilitate data sharing among participating firms and regulatory authorities.
Broader use of MDSAP audit reports by regulatory authorities, such as accepting these for a preapproval inspection, is also being considered.
According to Boyd, other FDA pilot programs available to industry that will play a role in the least burdensome initiative include the Case for Quality (CfQ) pilot and the Premarket Approval (PMA) CfQ pilot, where agency staff will seek to identify how to implement a more collaborative approach for resolving issues flagged during site inspections, and streamline the PMA pathway with earlier engagement on manufacturing reviews and processes.
Turbotax for device submissions
CDRH Chief Medical Officer William Maisel also briefly discussed the opportunity to move toward global regulatory convergence by harnessing the potential of electronic submissions. FDA committed to working on esubmissions as part of the latest reauthorization of MDUFA.
“The ideal model is Turbotax for device submissions,” Maisel said at the MedCon conference.
According to Maisel, CDRH developed a reviewer tool to begin piloting some esubmissions with a system in which questions from FDA guidance are built into a smart template to help ensure these are addressed.
"We're preparing a voluntary pilot where [firms would] be able to provide an electronic submission in a format similar" to the one being used by agency review staff, Maisel said.
The industry pilot would be designed so that “certain portions of an electronic submission can be shared across regulators and there may be additional modules specific to a given jurisdiction," Maisel added.
The idea is for this to ultimately help cut down on agency review time because it will allow for quality submissions and these would be formatted in a way that would make it more productive for the agency to review.
Boyd told Focus the new system could be expected to work similarly as eSubmitter—the software used for CDRH's Electronic Medical Device Reporting (eMDR) system to prepare submissions to FDA's main gateway. eMDR and eSubmitter have already been receiving simultaneous coding updates for harmonization with IMDRF terminology.

