OND Reorganization Moves Forward
The US Food and Drug Administration said Thursday that the planned reorganization of its Office of New Drugs (OND) will move forward thanks to Congressional approval, including changes to its Office of Translational Sciences (OTS) and the Office of Pharmaceutical Quality (OPQ).The changes will create offices for interrelated disease areas and divisions with clearer and more focused expertise, FDA said, noting the number of OND offices that oversee its review divisions will increase from six to eight, and there will be increases in the number of OND clinical divisions from 19 divisions to 27, plus six non-clinical review divisions.
“I am proud to say that the reorganization aspect of the New Drugs Regulatory Program modernization effort has cleared Congress, and we are now entering the next phase of our plan,” said Janet Woodcock, director of FDA’s Center for Drug Evaluation and Research.
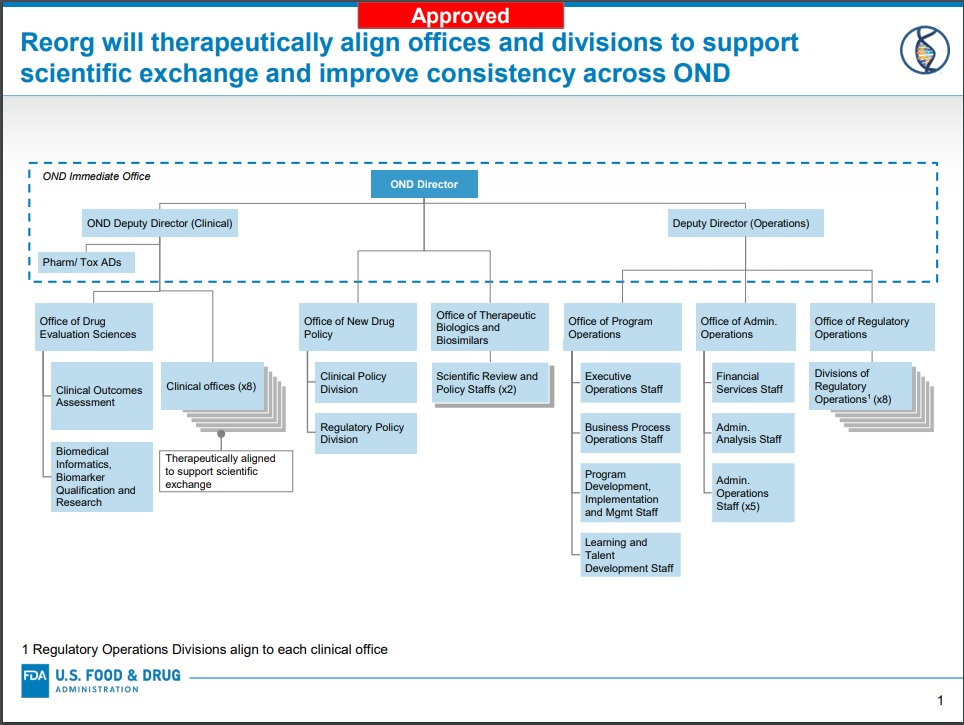
Previously discussed at last year’s RAPS’ Regulatory Convergence by Woodcock, the reorganization was first announced in June 2018 and OND’s more than 1,000 staffers will work in a more cross-disciplinary way, she said.
“Previously, CDER reviewers would seek consults from specialists in other scientific disciplines (as issues were identified in the course of review). For greater collaboration, a cross-disciplinary team will be assigned to work on a new drug application at the outset,” Woodcock wrote.
FDA also said that the changes include establishing the new Offices of Program, Administrative, and Regulatory Operations; an Office of New Drug Policy; and an Office of Drug Evaluation Sciences.

