Introducing RAPS Library, a new way to unlock expert regulatory knowledge
RAPS books help regulatory professionals improve their knowledge and find important information quickly.
Whether you’re a student looking to join the profession, a regulatory veteran, or someone who works in a field adjacent to regulatory affairs, our expert authors make our books for you.
Today, we’re excited to introduce RAPS Library, a new technology that makes RAPS’ expert-written books more accessible. We’re also debuting new subscription options to let regulatory professionals browse purchased books with ease, with the assurance they are accessing the most updated information available.
How RAPS Library helps you
Our books are still written by regulatory experts who take the time to make resources that propel the profession forward, building on shared knowledge to provide you with the information you need to bring safe, effective products to the public.
That isn’t changing.
What is changing is how you can access these books.
Search inside our regulatory books — before and after you buy
In RAPS Library, all RAPS books are now fully searchable. Once you purchase a book through RAPS Library, our new technology makes it even easier to search through that publication in a pinch when you need to find specific information quickly.
For example, if you are looking for information on digital health or artificial intelligence, RAPS Library lets you search our entire library to see which books have the materials you are looking for.
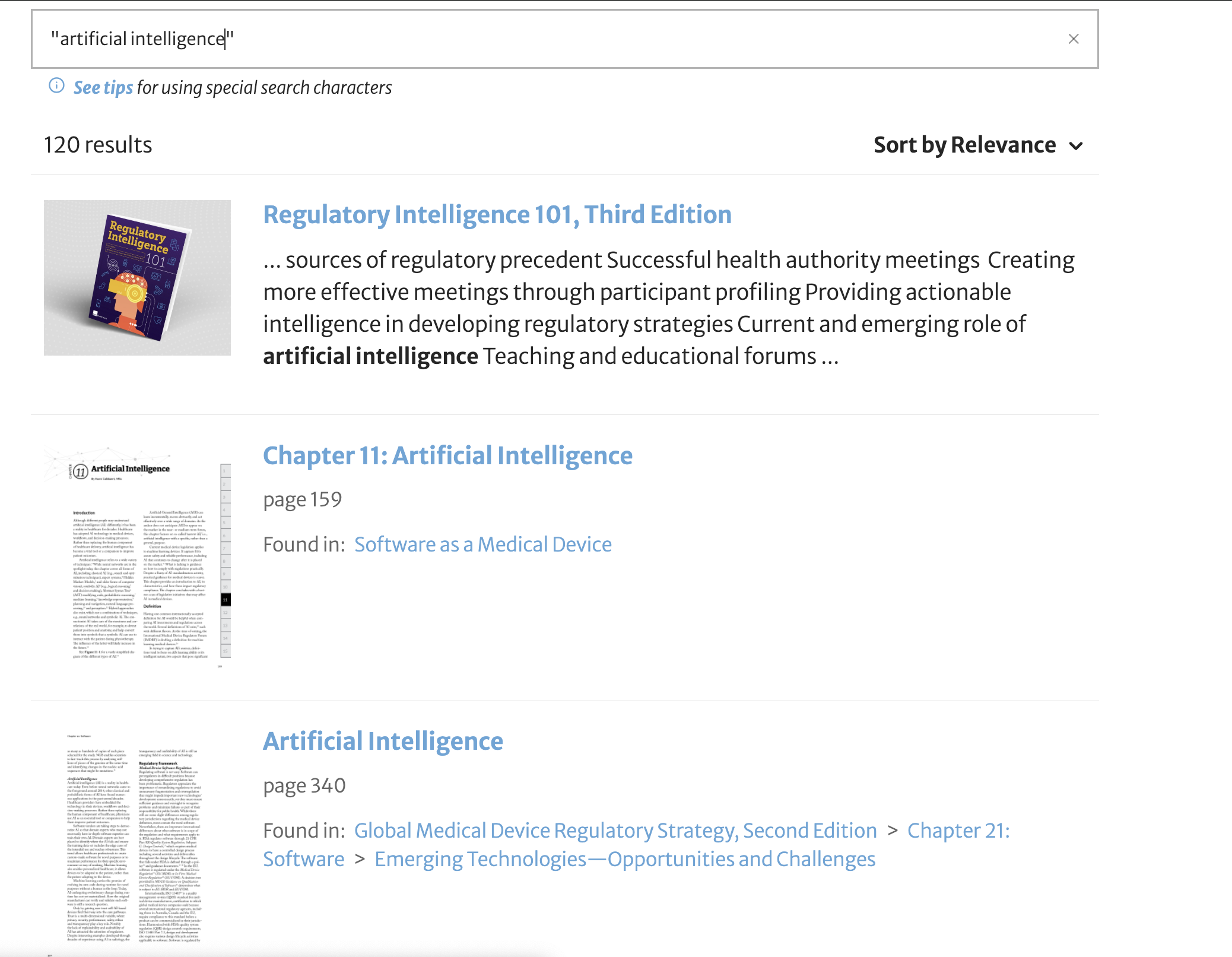
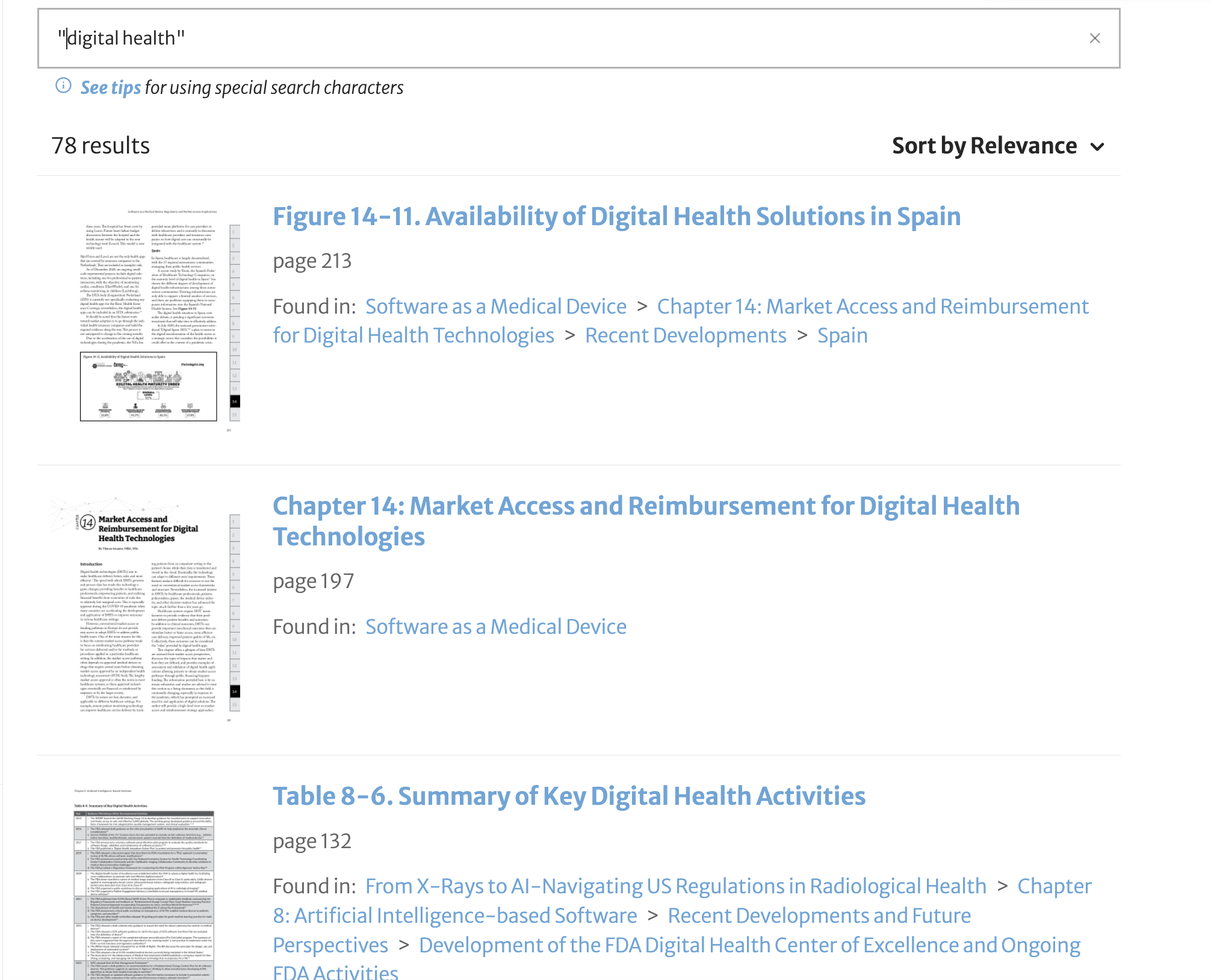
These are just a few examples. Try it yourself! Search the RAPS books catalog at library.raps.org.
Note: while this new technology does let you search our books, it does not let you read books for free. If you click on a search result but have not purchased the book, or if you are not logged in, you will receive this prompt.
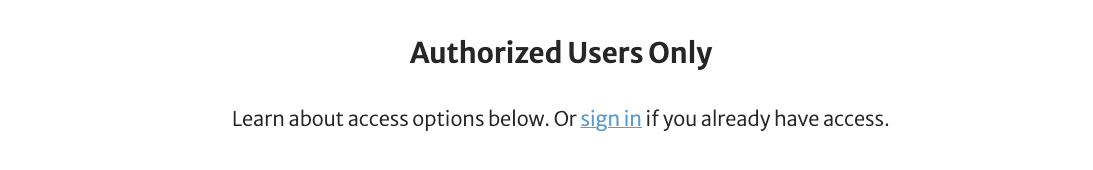
E-book subscriptions to fit your career
Our new technology allows RAPS to offer products previously unavailable to the regulatory profession. We’re debuting this technology with four e-book subscriptions designed with regulatory professionals in mind. These subscription options will let you spend less time searching for information and more time utilizing that information.
Your purchase will include any revisions made to our books during the lifetime of your subscription, so you’ll browse knowing you’re using the most updated resource the regulatory profession has to offer.
1. RAPS Fundamentals Excellence Bundle
Included:
-
Fundamentals of Pharmaceutical and Biologics Regulations: A Global Perspective
-
Fundamentals of Medical Device Regulations, Fifth Edition
Our two Fundamentals books combine to give you a comprehensive guide to regulatory requirements and rationales for pharmaceuticals, biologics, medical devices, in vitro diagnostics, and digital health products. Get unparalleled insights and expertise from more than 90 contributing authors at your fingertips.
2. Regulatory Excellence Bundle: Drugs and Biologics
Included:
-
Fundamentals of Pharmaceutical and Biologics Regulations: A Global Perspective
-
Global Pharmaceuticals and Biologics Regulatory Strategy, Second Edition
-
Regulatory Intelligence 101, Third Edition
For professionals who want to focus on drugs and biologics, our comprehensive Fundamentals book lets you research and reference regulations and guidance around the world, while books covering global regulatory strategy and regulatory intelligence allow you to thoroughly explore those topics.
3. Regulatory Excellence Bundle: Medical Devices
-
Fundamentals of Medical Device Regulations, Fifth Edition
-
Global Medical Device Regulatory Strategy, Second Edition
-
Regulatory Intelligence 101, Third Edition
For professionals who want to learn about medical devices, our comprehensive Fundamentals book lets you research and reference regulations and guidance around the world, while books covering global regulatory strategy and regulatory intelligence allow you to thoroughly explore those topics.
4. All Access Library
All Access Library customers get unlimited access to the entire RAPS books catalog. With expert writing from regulatory authors, you’ll be able to explore broad-based and narrow topics and automatically get updates as new editions are published and new titles are added to our catalog.
Learn more about our new e-book subscription products here.
Your expert regulatory resources, when and where you need them
Start browsing RAPS Library today to see how our expert-written regulatory books can help you.
