FDA leaders detail reorg plans, say 1,500 ORA staff will be reassigned
Top US Food and Drug Administration (FDA) officials have outlined the agency’s plans to reform its human foods program and transform its Office of Regulatory Affairs (ORA) into the Office of Inspections and Investigations (OII). They noted that about 1,500 ORA staff would be reassigned to product centers to work directly on inspections and investigations as part of the proposed reorganization.
On 19 December, the Alliance for a Stronger FDA (Alliance) hosted a webinar with Principal Deputy Commissioner Janet Woodcock, Chief Scientist Namandjé Bumpus, Associate Commissioner for Regulatory Affairs Michael Rogers, Deputy Commissioner for Human Food Jim Jones, and Donald Prater, acting director of the Center for Food Safety and Applied Nutrition (CFSAN). The officials discussed the agency’s proposal to reform its human foods program and ORA following criticism from Congress of its handling of the 2022 infant formula crisis.
Woodcock, who is leaving the agency next month, said that it’s taken a long time to get the ball rolling on the reorganization proposal due to bureaucracy, but that officials are hopeful the reorganization will be implemented this year after it’s been agreed to by FDA’s unions. She added part of the goal is to stop duplicating functions at ORA and noted that the plan is budget neutral.
“We're really trying to transform different aspects of the FDA to make them more efficient, more effective, and so forth,” said Woodcock. “Our mission seems to continually be broadening and we really do need to be able to meet our mission as much as our resources allow.”
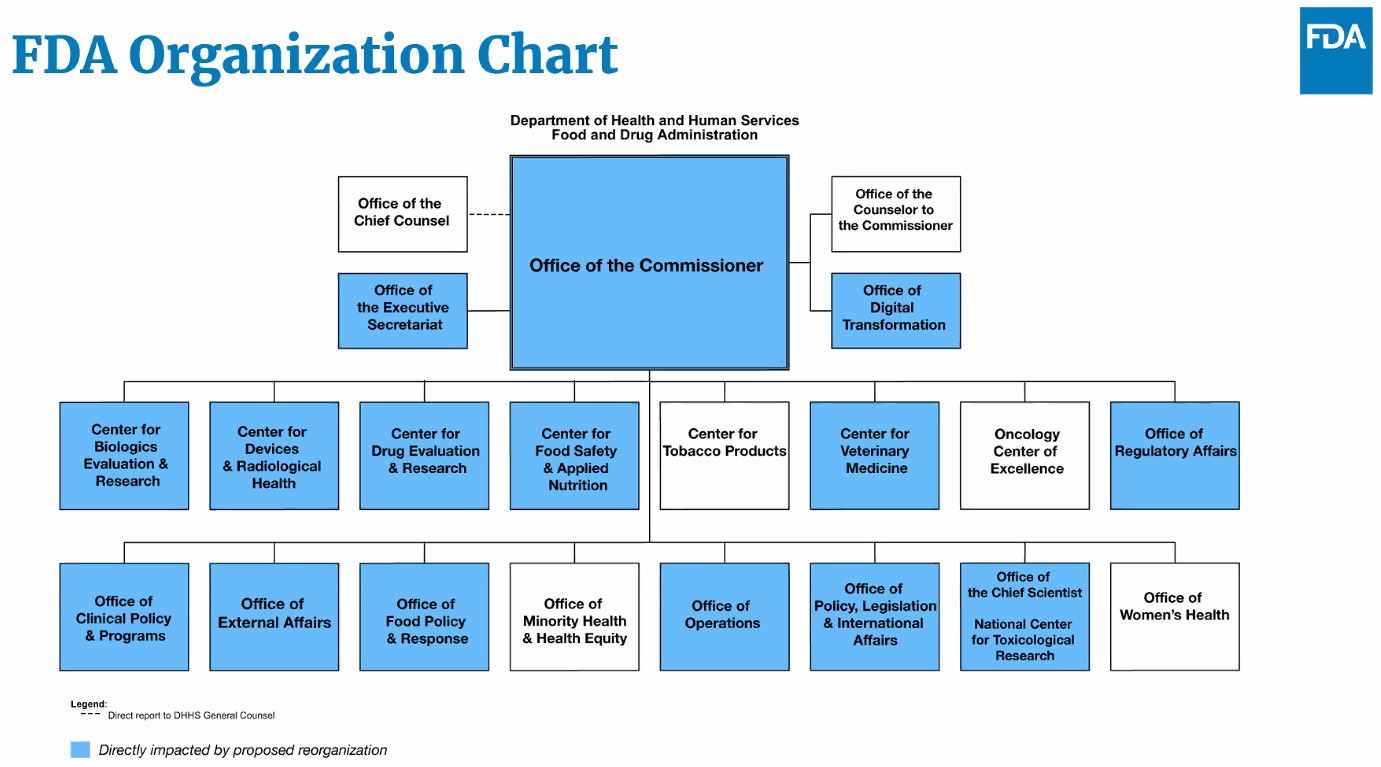
During the webinar, Woodcock noted FDA submitted the reorganization plans to the Department of Health and Human Services (HHS) last month and added that in the future the agency will be more transparent about how their budget is being spent. She also presented an organizational chart that showed that most FDA offices will be affected in some way by the reorganization.
During the webinar, Rogers spoke about how ORA would be transformed as OII, and noted it will be specifically focused on inspections, investigations, and import operations. He added that most compliance functions that were under ORA, except for imports, would be reassigned to the product centers and compliance programs that are already established in those centers.
“That alignment will eliminate a lot of duplication and bring investigators closer to the center program staff, especially when the agency needs to react to and evaluate an ongoing violative inspection,” said Rogers. “In addition, most consumer complaint responsibilities in ORA, specifically intake and evaluation and closeout, will be realigned to the human foods program and the centers, and OII will carry out any necessary field evaluation for complaints when requested by the human foods program in the centers and will also retain a small team of complaint personnel to make sure these field evaluations are conducted in a streamlined manner.”
He said that this will mean ORA staff would shrink from about 5,100 to 3,600 at OII. The remaining 1,500 staff would be reassigned to the agency’s product centers.
Rogers argued the reorganization will enable FDA to track product complaints during their lifecycle better, ensure complaints are handled in a timely manner, and serious complaints are sent up the chain of command to senior leadership to address. He also noted that while the human food laboratories will be assigned to the human foods program, other product laboratories will be assigned to the Office of Chief Scientist.
Rogers also presented a chart of how OII will operate and noted that there will be several cross-cutting functions that it will be responsible for.
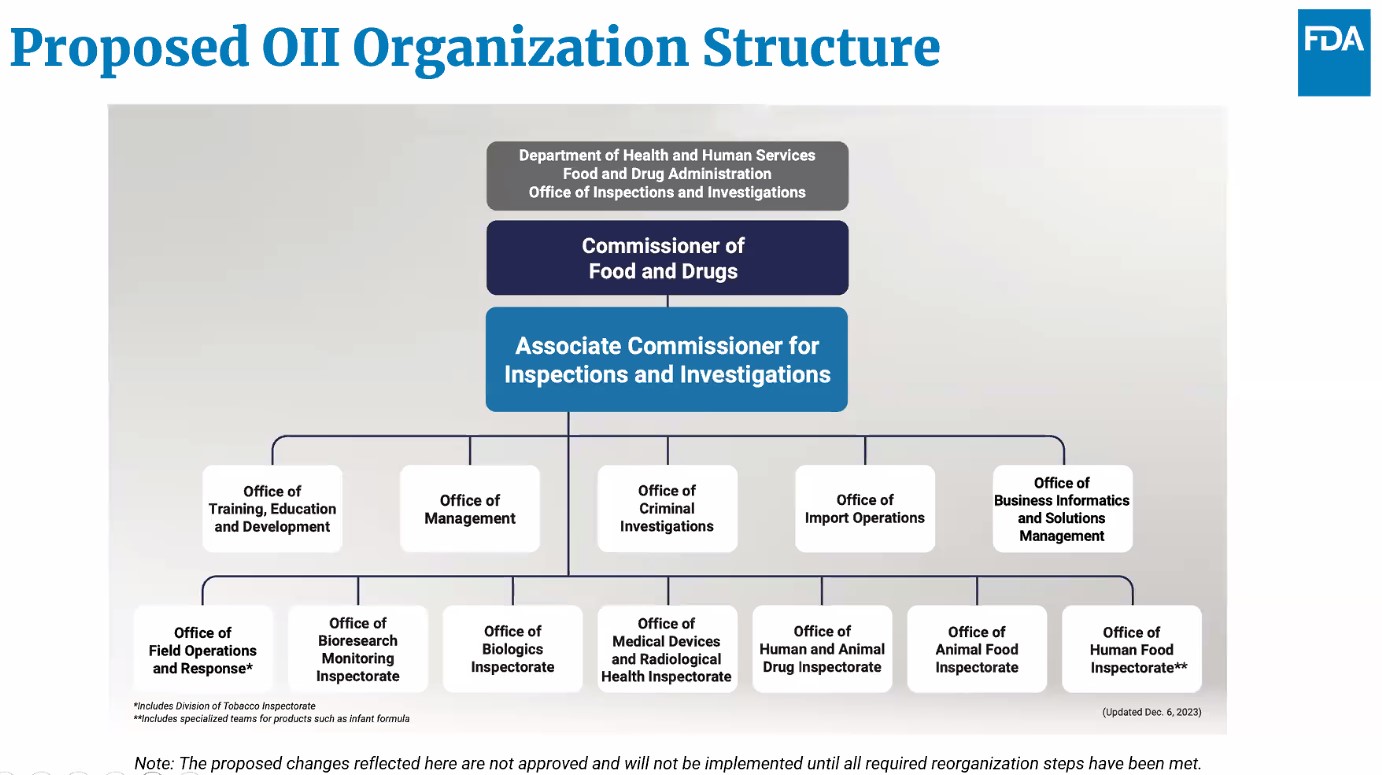
“For example, our Office of Criminal Investigations remains essentially unchanged and will carry out criminal investigation activities for all of the FDA-regulated products,” he said. “We’ll have an Office of Field Operations and Response that will provide enterprise inspectorate support, including organizational quality efforts and within it there'll be a division of tobacco inspectorate to conduct tobacco inspectional work.”
Rogers also added that his office will also have an Office of Emergency Response that will coordinate FDA’s response to emergencies and natural disasters that are related to FDA-regulated products and FDA facilities with the exception of foodborne outbreaks, which will be managed by the human food program. He noted that the responsibility was previously under the Office of Operations.
During the webinar, Bumpus noted that after HHS has reviewed the proposed reorganization plan, it will be sent to the Office of Management and Budget (OMB) for review, Congress will receive a 30-day notification period, a Federal Register notice will be published that will allow stakeholders to comment on the plan, and FDA will begin negotiations with staff unions.
“FDA is hopeful that implementation will occur sometime in calendar year 2024,” she added.
Now that they’ve presented their reorganization plans to HHS, Woodcock said the agency is working out the details to ensure the transition happens smoothly.
“There will be an initial period of adjustment as the different staff who are moving around, working in slightly different ways, have to adjust to that should we be able to stand up the reorg this year,” she said, adding that she thinks the transition will go smoothly.
Video of Alliance webinar




